The f-block elements appears as a footnote of two series in a standard 18-column table but is located at the center-left of a 32-column full width table. They are sometimes called inner transition metals because they provide a transition between the s-block and d-block in the 6th and 7th periods. All are metals. The f-orbital electrons are largely inactive in determining the chemistry of the 6th period f-block elements. Their chemical properties are mostly determined by a single d and two s-orbital electrons.
The f-block elements are unified by mostly having one or more electrons in an inner f-orbital. Of the f-orbitals, five have six lobes each, and the sixth looks like a dumbbell with a donut with two rings. They can contain up to seven pairs of electrons hence the block occupies fourteen columns in the periodic table.
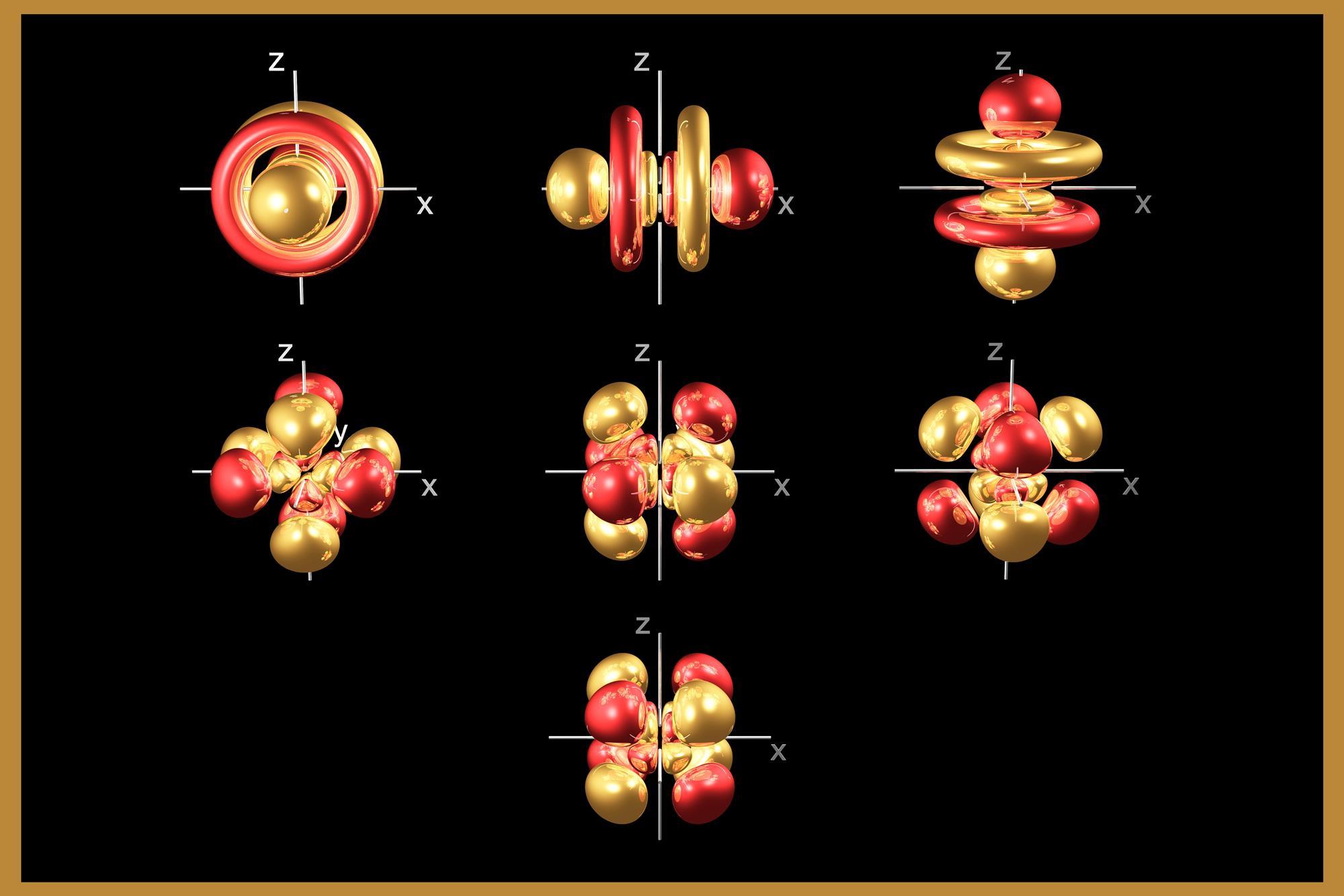
The two 14-member rows of the f-block elements are sometimes confused with the lanthanides and the actinides, which are names for sets of elements based on chemical properties more so than electron configurations. The lanthanides are the 15 elements running from lanthanum (La) to lutetium (Lu); the actinides are the 15 elements running from actinium (Ac) to lawrencium (Lr).
- Lanthanides
The lanthanide (or lanthanoid) series is a series of 15 metallic chemical elements that have atomic numbers 57 to 71. This group starts from lanthanum and it ends at lutetium. The lanthanoid series (Ln) is named after lanthanum because the elements in the series are chemically similar to lanthanum. All lanthanoids are f-block elements and has the electronic configuration as 5d¹ 4f¹⁻¹⁴, which means that electrons fill the 4f electron shell, except for lanthanum and lutetium which is a d-block lanthanoid. All lanthanide elements form trivalent cations (3⁺ ions), whose chemistry is largely determined by the ionic radius, which decreases steadily from lanthanum to lutetium. Both lanthanum and lutetium have been labeled as group 3 elements, because they have a single valence electron in the 5d shell.

Lanthanoids are metals that are related to each other in how they behave. Most of them slowly turn into their hydroxides when they are placed into water, like the alkali metals. They usually form a coating of oxide when placed in the air. Lanthanides with Scandium and Yttrium are called rare earth elements. All the lanthanoids are silvery white soft metals and turn tarnish rapidly in air. The hardness increases with increasing atomic number. Lanthanides are not abundant, and only small amounts are mined. Some have various uses in magnets, superconductors, chemical catalysts, and optical equipment such as lasers.
- Actinides
The actinide series is a series of 15 metallic chemical elements that have atomic numbers from 89 to 103. This group starts from actinium and it ends at lawrencium. The actinide series derives its name from the first element in the series, actinium. All of the elements are radioactive. Uranium is the most common natural actinide, and thorium second.
All of the actinides mostly corresponds to the filling of the 5f electron shell. Hence, they are f-block elements, with the exception being either actinium or lawrencium. Both actinium and lawrencium have been labeled as group 3 elements. In comparison with the lanthanides, also mostly f-block elements, the actinides show much more variable valence. They all have very large atomic and ionic radii and exhibit an unusually large range of physical properties. While actinium and the late actinides (from americium onwards) behave similarly to the lanthanides, the elements thorium, protactinium, and uranium are much more similar to transition metals in their chemistry, with neptunium and plutonium occupying an intermediate position.
All actinides are radioactive and release energy upon radioactive decay; naturally occurring uranium and thorium, and synthetically produced plutonium are the most abundant actinides on Earth. These are used in nuclear reactors and nuclear weapons. Uranium and thorium also have diverse current or historical uses, and americium is used in the ionization chambers of most modern smoke detectors.



Comments
Post a Comment